[Study News] Phase 1 clinical trial of VectorFlu ONE, COVID-19 nasal vaccine, has commenced!
14/05/2021

[Study News] Phase 1 clinical trial of VectorFlu ONE, COVID-19 nasal vaccine, has commenced!
14/05/2021
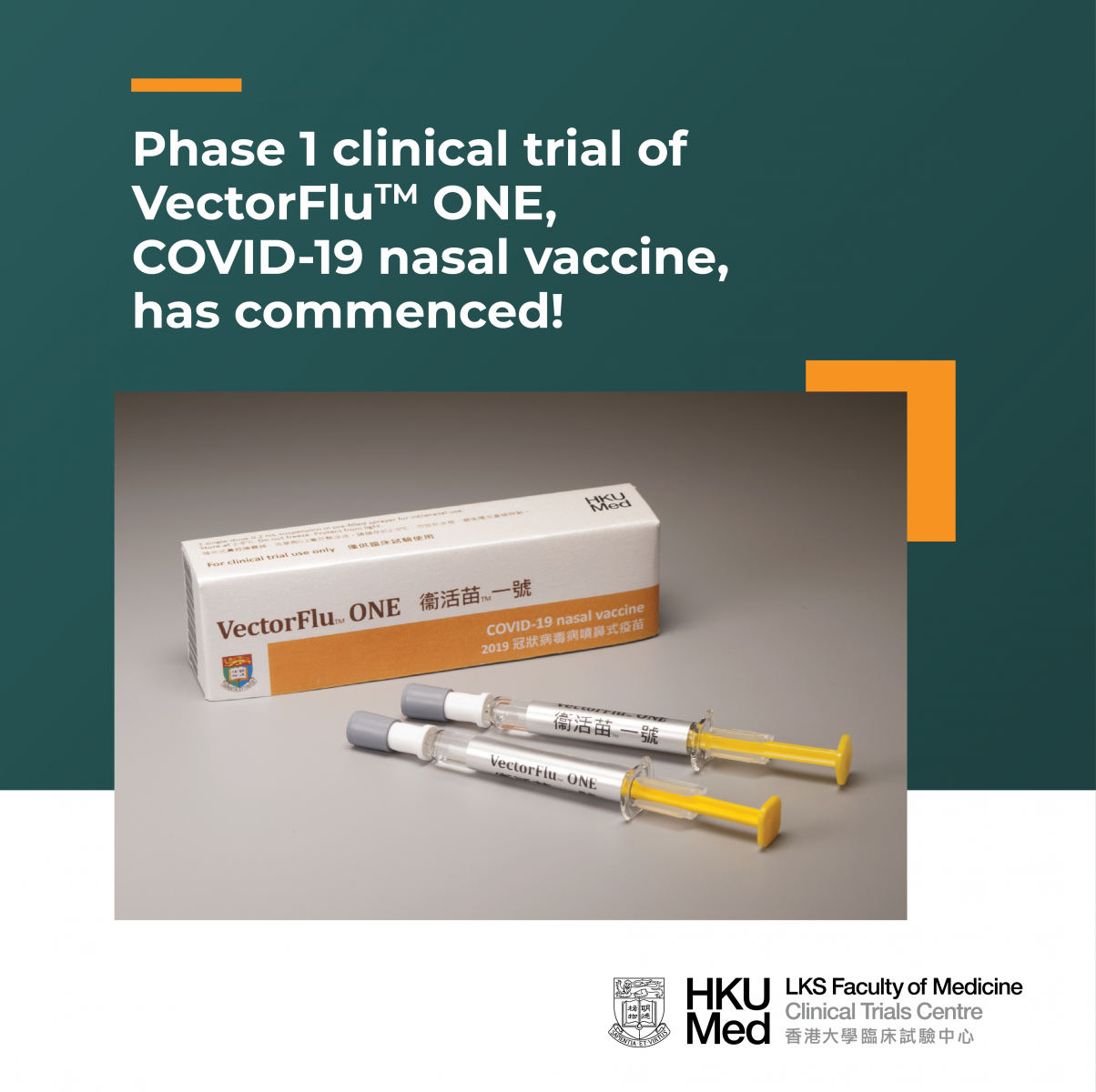
The phase 1 clinical trial of VectorFluTM ONE, the world’s first COVID-19 nasal vaccine developed by the research team of HKU Department of Microbiology, has commenced in March 2021 at the HKU Phase 1 Clinical Trials Centre. About 115 healthy volunteers will be recruited to evaluate the safety and immune responses of the vaccine.
VectorFluTM ONE is the first novel vaccine that is developed by Hong Kong and goes into human trial. The phase 1 trial will last for around 1.5 years. Part 1 of the trial has commenced in March 2021, where about 30 volunteers have already received one or two vaccinations. Part 2 will begin in July 2021 and 85 volunteers will be recruited. We are still looking for more volunteers to join.
Successful Phase 1 Study on Novel FcRn Inhibitor Offers New Direction for Treatment of Autoimmune Disease
04/05/2021
Successful Phase 1 Study on Novel FcRn Inhibitor Offers New Direction for Treatment of Autoimmune Disease
04/05/2021
[Pharma Tips] Vaccine Technology for COVID-19 at a Glance
26/02/2021
Formulation of effective vaccine strategies for COVID-19 is not straight forward as the immune responses to COVID-19 in humans are still unclear. Application of various technologies would be necessary for developing safe and effective vaccines, as different vaccine technologies may be needed for different population. Immediately after the release of the genome sequence of SARS-CoV-2 in January 2020, multiple vaccine technologies have been investigated and proceeded to different stages of pre-clinical and clinical trials.
Below are the current major vaccine technologies for COVID-19 and their respective mechanisms:
Immune Response
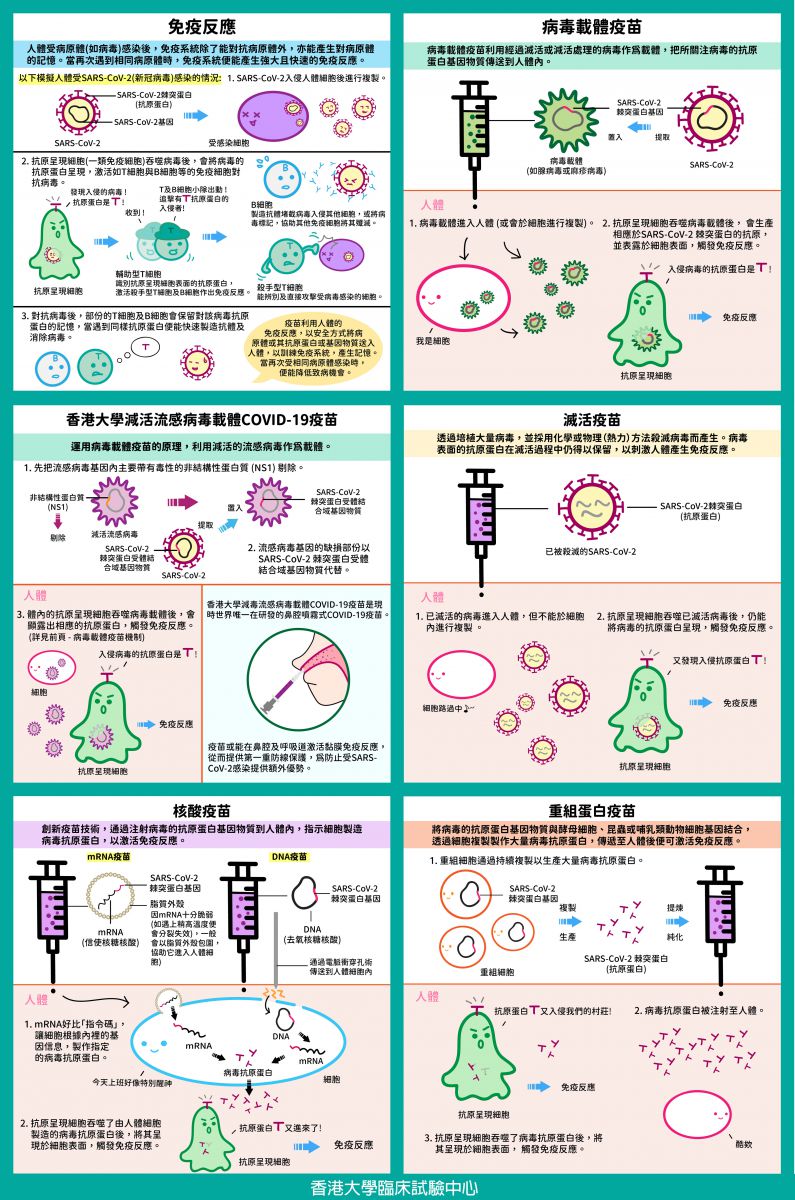
When the human body is infected by a pathogen (e.g. virus), the immune system may fight the pathogen and develop memory towards it. When the same pathogen is encountered again, the immune system may establish strong and rapid immune responses.
By utilizing the human immune responses, vaccines may train the immune system to recognize a pathogen by safely delivering the pathogen or its antigen or genetic material into the human body. Therefore, when the human body is exposed to the same pathogen again, the chance or severity of infection may be lowered.
5 Types of Vaccine Technology
Viral Vector Vaccine
Viral vector vaccine uses a virus without virulence as a vehicle to deliver the genetic material of the antigens of the virus of interest into human body. The genetic material of the viral antigens uses human cells’ machinery to produce the respective antigens and presented on the cell surface, the immune system will then react.
Viral vector vaccine has been extensively studied to fight against infectious diseases, including influenza, HIV and Ebola etc. The most common viral vector platform used for vaccine production is adenoviral vector. A recently approved COVID-19 viral vector vaccine by regulatory authorities uses adenovirus as vehicle to deliver the genetic material of the spike protein of SARS-CoV-2 into human cells, at where the spike protein is expressed, and trigger the immune system to produce antibodies and T-cell responses.
HKU’s Flu-based Live Attenuated Viral Vector COVID-19 Vaccine
The HKU’s flu-based live attenuated viral vector vaccine uses attenuated influenza virus as the viral vector. Its key virulent element, non-structural protein NS1 is removed which makes the influenza vector severely attenuated, and this knock-out region is replaced by the genetic material of the receptor binding domain (RBD) of SARS-CoV-2 spike protein. The RBD of SARS-CoV-2 spike protein will eventually trigger the immune response following the above viral vector vaccine mechanism.
HKU’s flu-based live attenuated viral vector COVID-19 vaccine is the only intranasal COVID-19 vaccine under development worldwide. It might have additional benefit of inducing mucosal immune response in the nasal cavity and respiratory tract, and offer a first line protection against SARS-CoV-2 infection. The phase 1 clinical trial of this vaccine will be conducted in HKU Phase 1 Clinical Trials Centre, and is under preparation by Department of Microbiology, Department of Medicine and Clinical Trials Centre of the University of Hong Kong.
Inactivated Virus Vaccine
Through cultivation of a large amount of viruses and killing the viruses by chemical or physical (heat) technology, inactivated virus vaccine is produced. Immune responses are triggered by the preserved antigens presented on the viral surface. Since inactivated viruses could not replicate by themselves, the quantity of virus required will be large, which may result in higher dosage level and multiple vaccinations. This vaccine production technique has been long established and been applied to vaccines against influenza, polio and rabies etc. Currently, some COVID-19 vaccines approved by regulatory authorities have adopted this inactivated virus vaccine technique.
Nucleic Acid Vaccine
Nucleic acid vaccine against infectious diseases is an innovative vaccine technology. Currently, mRNA (messenger RNA) and DNA vaccine technologies are under active development worldwide against COVID-19. Both technologies aim at triggering immune responses through injecting genetic materials coded for the spike protein of SARS-CoV-2 into human bodies.
mRNA vaccine: Encased in a lipid coat, mRNA carrying the genetic information of viral antigens is injected into human body, where it instructs ribosomes to synthesize viral antigens. Immune responses are then triggered.
DNA vaccine: The DNA plasmid with the genetic information of the viral antigens is transmitted into human cells through electroporation, and then is translated into viral antigens through mRNA. Immune responses are then triggered.
Recombinant Protein Vaccine
The genetic materials of the viral antigens are delivered into yeast cells, insect (baculovirus) or mammalian cells to express viral antigens. These recombinant cells continue to multiply and produce large quantity of viral antigens. The viral antigens will be extracted from the cells and purified, of which recombinant protein vaccine is produced.
Immune responses will be triggered by the viral antigens once the vaccine is injected into human body. This technology has been adopted for producing Hepatitis B vaccine and Human Papillomavirus vaccine. Through the expression of antigens of SARS-CoV-2 (e.g. spike protein), recombinant protein vaccine could be developed for fighting against COVID-19.
[Study Completion] Successful Completion of Generic Drug Study Indicated for Hyperlipidemia!
30/12/2020

[Study Completion] Successful Completion of Generic Drug Study Indicated for Hyperlipidemia!
30/12/2020
The recruitment of this study started in July 2020 and 52 healthy volunteers were recruited. This clinical study consisted of 2 parts (fasted and fed), and each volunteer only joined either one part. The duration of each part was around 4-6 weeks, including screening, confinement and follow up visits. Although the study process was slightly postponed due to the COVID-19 pandemic, thanks to the genuine support from our volunteers and the effort of our HKU professional research team, the project was completed safely and efficiently in November 2020.

[Study Completion] Phase 1 Clinical Trial of a New Autoimmune Disease Drug Was Completed!
01/12/2020

[Study Completion] Phase 1 Clinical Trial of a New Autoimmune Disease Drug Was Completed!
01/12/2020
HKU Clinical Trials Centre was delegated to research on a novel drug for treating autoimmune diseases. Phase 1 trial of the drug was conducted in Canada earlier but the research purpose was to evaluate the metabolism and safety of the drug at dose escalation in Chinese population.
Recruitment started in May 2019 and 24 qualified healthy volunteers were selected after screening. The 18-week research was divided into 3 cohorts and each volunteer only joined one of them. The volunteers received injection during their 6-day-5-night stay in the Centre. As the duration of overnight stay was long, two workshops were organized, namely Spark Chamber and Mini Neon Light DIY Workshops.
The clinical study was successfully completed in November 2020.

[Centre Activity] The 6th International Clinical Trial Center Network (ICN) Steering Board Meeting and Annual General Meeting 2020
27/11/2020
[Centre Activity] The 6th International Clinical Trial Center Network (ICN) Steering Board Meeting and Annual General Meeting 2020
27/11/2020
HKU-CTC successfully hosted the 6th ICN Steering Board Meeting and Annual General Meeting 2020 on 24-25 November, 2020. Participants engaged actively and had vivid discussions – even though the meetings were moved online due to the pandemic.
At the Steering Board Meeting, Mr. Henry Yau, Managing Director of HKU-CTC, was elected the new ICN Chairperson. He will work with the new Vice Chairperson, Dr. Christiane Blankenstein of Munich Study Center, Technical University of Munich, the ICN Steering Board, and the ICN Operations Team to contribute to the further development of ICN in the coming two years. The Steering Board also formulated a strategic plan towards realization of the values of “Global, Excellence, Harmonization, Impacts”.
The next ICN Steering Board Meeting and Annual General Meeting will be hosted by Cambridge Clinical Trials Unit (CCTU), Cambridge University Hospitals NHS Foundation Trust in November 2021.

Combined Use of Immunotherapy Drugs Improves Clinical Outcomes for Patients with Advanced Hepatocellular Carcinoma and Received FDA Approval as a Second-line Therapy for HCC
10/11/2020
Combined Use of Immunotherapy Drugs Improves Clinical Outcomes for Patients with Advanced Hepatocellular Carcinoma and Received FDA Approval as a Second-line Therapy for HCC
10/11/2020
[Centre Activity] PRACTISE® IIS - Training Course for Investigator-Initiated Clinical Studies
31/08/2020

[Centre Activity] PRACTISE® IIS - Training Course for Investigator-Initiated Clinical Studies
31/08/2020
Clinical research is an essence of medical advancement and a cornerstone of a robust health care system. In general, there are two types of clinical research – industry-sponsor studies and investigator-initiated studies (IISs) – whereas those initiated by investigators are increasingly important nowadays for its variety and diversity. They also bridge the research gaps on areas which are rarely covered by the industry. Facilitation of IISs is therefore a focus of The University of Hong Kong Clinical Trials Centre (HKU-CTC). In order to encourage investigators to initiate more high impact IISs, HKU-CTC has recently launched PRACTISE® IIS – a training programme addressing the specific considerations for IISs.
The first course was completed by way of an online seminar on 28th August, 2020. Professor Ian Wong, Head of Department of Pharmacology and Pharmacy, and Dr. Helen Zhi, Director of Biostatistics and Clinical Research Methodology Unit, shared their practical tips and experience in designing and managing big data research and clinical studies with over 300 participants and received enthusiastic response. CTC will continue to organize other courses on IISs.